Abstract
Introduction Anti-CD38 antibody-based therapies in multiple myeloma (MM) rely to a large degree on natural killer (NK) cell-mediated antibody-dependent cellular cytotoxicity (ADCC). Classically, NK cells are divided into cytotoxic CD56dim NK cells, important for ADCC, and cytokine-producing CD56bright NK cells. Accumulating evidence in other types of cancer suggests that alterations in the balance between CD56dim and CD56bright NK cells is associated with inferior clinical outcome. We hypothesized that changes in NK cell subset composition in the MM microenvironment would impact therapy-responses during NK cell-driven immunotherapies. Here, we used single-cell RNA sequencing and flow cytometry to investigate the heterogeneity of the NK cell compartment in the bone marrow (BM) of newly diagnosed MM (NDMM) patients, and the effects of altered NK cell subset composition on therapy response in patients undergoing first-line daratumumab-containing therapy.
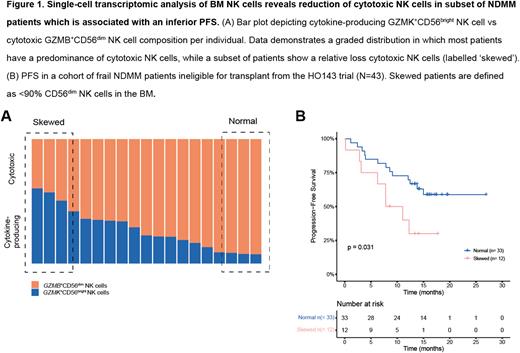
Results We performed single-cell RNA sequencing of the BM CD38+ immune cell compartment from 19 NDMM patients. NK cells were identified in silico resulting in a single-cell transcriptomic dataset of 24,664 NK cells. UMAP dimensionality reduction identified 5 transcriptomic distinct NK cell clusters, present in all NDMM patients. Conventional CD56bright and CD56dim NK cells were identified by increased transcription of GZMK or GZMB, respectively. To study the balance of cytotoxic vs cytokine-producing NK cells in MM we compared the relative abundance of cytokine-producing GZMK+CD56bright NK cells vs cytotoxic GZMB+CD56dim NK cells in individual patients in our cohort. The MM BM was characterized by heterogeneity in the ratio of cytotoxic vs cytokine-producing NK cell cluster composition, in which most patients had a predominance of cytotoxic NK cells, while a subset of patients presented with a relative decrease in cytotoxic NK cell clusters (Figure 1A).
The reduction in cytotoxic NK cell subsets in this patient subgroup was reflected in an altered overall transcriptome of the total BM NK cell population, with enrichment for inhibitory receptors such as KLRB1 and KLRC1, and a relative loss of activating receptors such as FCGR3A (CD16), NCR3 (NKp30) and CD226 (DNAM-1), and cytotoxic effector genes including NKG7 and GNLY.
To define the relative abundance of a reduced cytotoxic NK cell potential in NDMM patients, we performed flow cytometric analyses of 246 NDMM patients from 2 independent clinical cohorts. This revealed a reduced frequency of cytotoxic CD56dim NK cells, defined as < 90% CD56dim NK cells, in 17.9 % of 162 transplant-eligible treatment-naïve NDMM and in 28.6% of 84 treatment-naïve frail or unfit transplant ineligible MM patients. To test whether such relative decrease in cytotoxic NK cells would impact responses to daratumumab, we tested the association of BM NK cell composition with clinical outcome in the context of first-line daratumumab therapy in a cohort of frail NDMM patients ineligible for transplant from the HO143 trial (n=43). NDMM patients with reduced frequencies of BM cytotoxic NK cells presented with significantly shorter progression-free survival (PFS) (Figure 1B, PFS, p = 0.031, log-rank test), while OS was not impacted (p = 0.18, log-rank test). Univariate Cox regression analyses confirmed the correlation between reduced frequencies of CD56dim NK cells in the BM and significantly shorter PFS (hazard ratio (HR) 1.38; 95% confidence interval: 1.057 - 1.803; P = 0.018). Whether reduced frequencies of cytotoxic CD56dim NK cells affect outcome in transplant-eligible patients warrants additional study.
Conclusion Here we show that about 20% of NDMM patients have an altered BM NK cell composition with a relative reduction in cytotoxic NK cells. Such altered NK cell composition correlates with significantly shorter PFS following daratumumab-containing therapy in a cohort of frail NDMM patients. Our data suggest that defining BM NK cell composition could stratify patients and identify a subgroup less likely to benefit from therapies driven by NK cell-mediated ADCC.
Disclosures
D'Agostino:Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Advisory board; GSK: Membership on an entity's Board of Directors or advisory committees, Other: Advisory board. Gay:AMGEN: Membership on an entity's Board of Directors or advisory committees; BMS/Celgene: Membership on an entity's Board of Directors or advisory committees. van der Velden:Agilent: Other: laboratory services agreement ; Cytognos: Other: laboratory services agreement ; BD biosciences: Other: laboratory services agreement ; Euroflow: Patents & Royalties. Van De Donk:Cellectis: Research Funding; Servier: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Adaptive Biotechnologies: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Membership on an entity's Board of Directors or advisory committees, Research Funding; Roche: Membership on an entity's Board of Directors or advisory committees; Bayer: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Janssen Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees, Research Funding. Broijl:Amgen: Other: Advisory boards/honoraria; Janssen: Other: Advisory boards/honoraria; Sanofi: Other: Advisory boards/honoraria; Bristol Myers Squibb: Other: Advisory boards/honoraria. Sonneveld:Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees, Research Funding; Karyopharm: Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Membership on an entity's Board of Directors or advisory committees, Research Funding. Zweegman:Takeda: Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Sanofi: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; Oncopeptides: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal